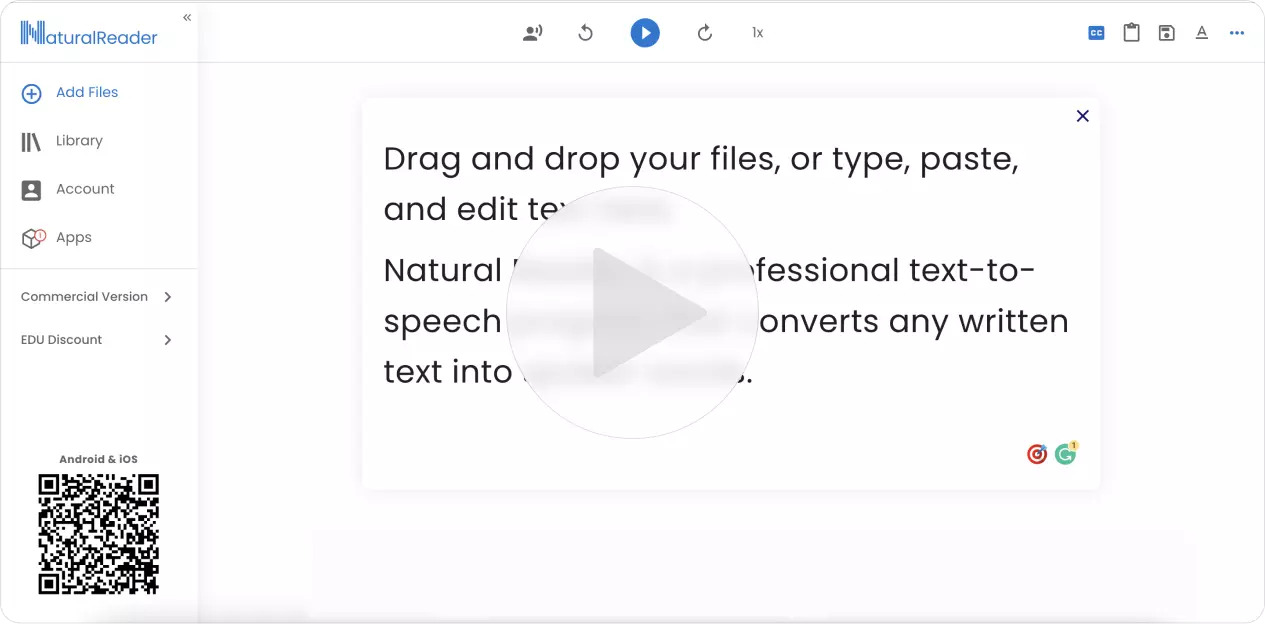

One account, all of NaturalReader
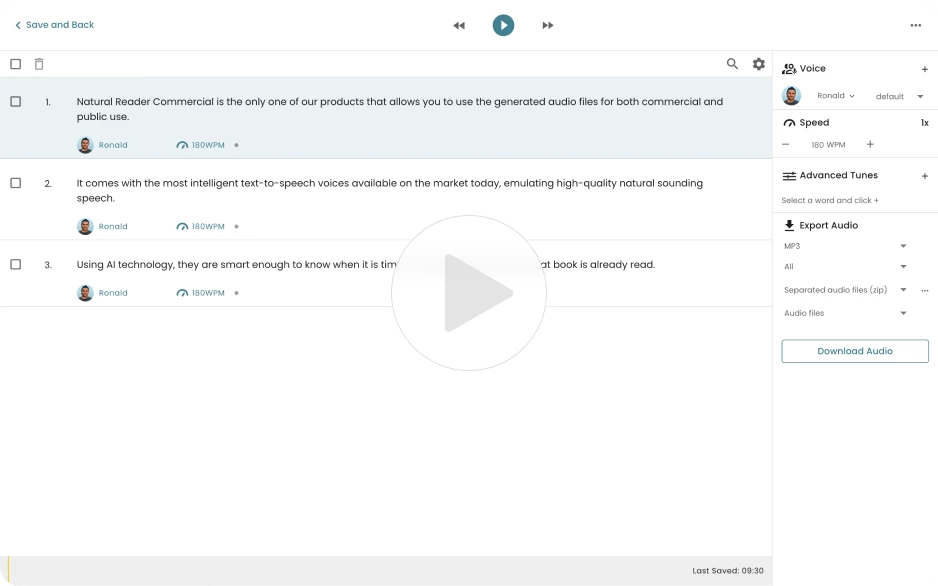
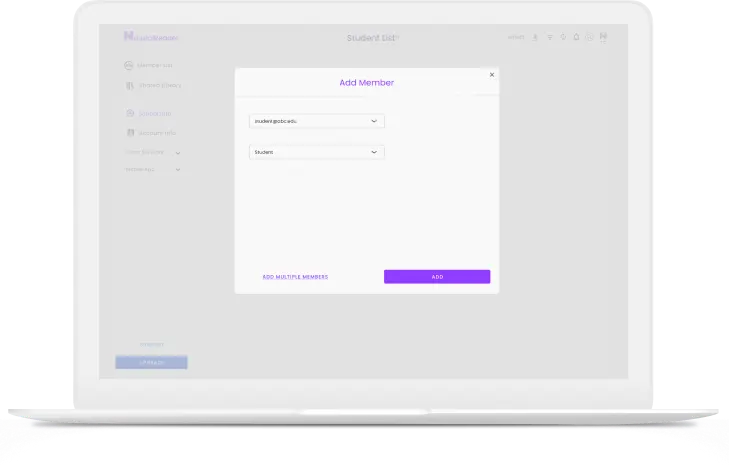
Add members through email or class code, share documents to a class, and manage or delete classes and members
Learn About EDUThe defining feature of an azo dye is the azo linkage connecting two aromatic fragments, typically aryl groups. This arrangement creates an extensive , which allows the molecule to absorb energy in the visible light spectrum (400–800 nm), resulting in intense, stable colors.
: The diazonium salt acts as an electrophile, reacting with an electron-rich nucleophile (like a phenol or another aromatic amine) to create the final azo bond. Industrial and Biological Applications
The synthesis of these dyes usually involves a two-step process:
: An aromatic amine reacts with nitrous acid at low temperatures to form a diazonium salt .